If you live in one of those “Red Zone” hard water districts — whether it’s the limestone-rich parts of the UK or the heavily mineralized aquifers in places like Texas and Florida — you probably know exactly how frustrating it can be to wash your face every single day. A lot of people in these areas keep noticing the same annoying problem: even the most expensive, rich, lipid-heavy moisturizers they buy just don’t seem to deliver the lasting hydration they promise. It feels like the product stops working halfway through the day, leaving your skin tight, dry, or even a bit flaky no matter how much you apply. The truth is, it’s rarely because the moisturizer itself is badly formulated. What’s really going on is the quiet buildup of a microscopic hard water skin film on the surface of your skin. This invisible layer forms through a natural chemical reaction called saponification — when the dissolved minerals in your tap water come into contact with the natural fatty acids and sebum that your skin produces every day. Over time, this hard water skin film creates a stubborn barrier that prevents even the best moisturizers from penetrating properly and locking in moisture the way they’re supposed to. It’s not your fault, and it’s not the product’s fault — it’s simply the hidden effect of living with hard water day after day.
Why Hard Water Leaves That Stubby Film on Your Skin?
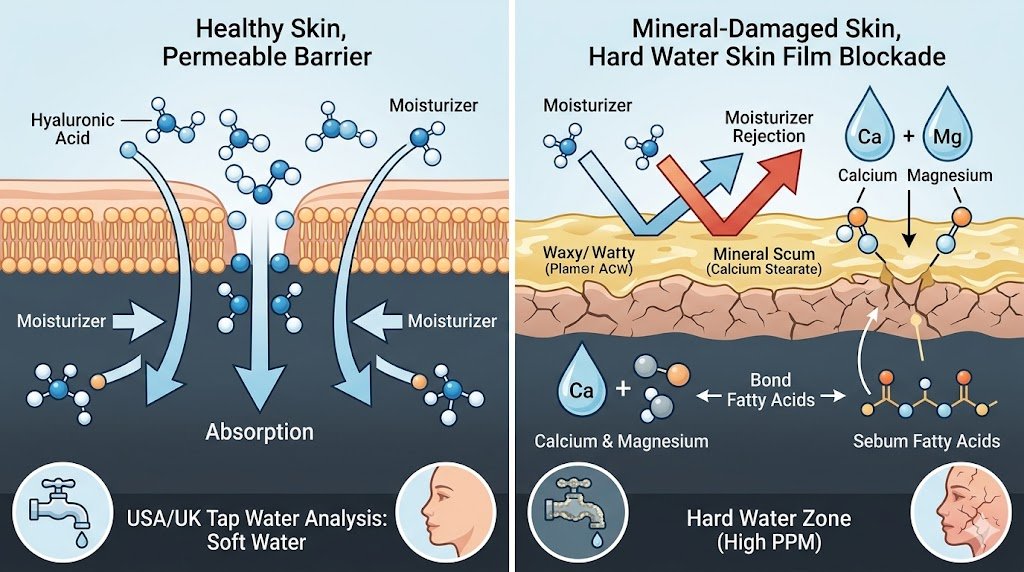
When you start looking at what’s really happening on your face in hard water areas, the chemistry behind that annoying “skin scum” on the dermis suddenly becomes a lot clearer. Every single time you wash your face, the minerals floating in your tap water — mainly calcium and magnesium — team up with the natural oils and fatty acids that your skin produces, like stearic acid and palmitic acid from your sebum. This reaction creates tiny insoluble salts, the same kind of stuff we all know as soap scum in the shower, technically called calcium stearate in the lab. The result is a thin, waxy, almost invisible hard water skin film that clings to your skin like a stubborn shield. This buildup comes from both sides: externally from all those heavy minerals constantly coming out of your taps, and internally from the steady flow of natural sebum and oils your own skin keeps making every day. Together they form this non-permeable layer that stops even the fanciest humectants and high-end moisturizers from sinking properly into the deeper layers of your skin where they’re meant to work. Instead of absorbing and giving you real, lasting hydration, everything just sits uselessly on the surface, leaving your skin feeling tight, dry, and disappointed no matter how much product you use.
If you want to dive deeper into the science of how hard water affects the skin barrier, you can read this detailed study on the effects of water hardness on skin here: The Effect of Water Hardness on Surfactant Deposition and Skin Barrier Function. And if you’re tired of fighting this exact issue and want real, practical ways to deal with hard water skin film.
The “Squeaky” Texture Reports (USA & UK Focus)
When you look through hundreds of user reviews and real skin stories from cities like London, San Antonio, and Chicago, the same pattern keeps showing up again and again. People often say their skin feels “squeaky clean” right after rinsing with tap water, but then within just ten minutes they notice an uncomfortable tightness and that dull, ashy look that makes their face feel dry and uncomfortable. That “squeaky” feeling isn’t actually a sign of deep cleanliness like many of us were taught to believe. Instead, it’s the physical sensation of the hard water skin film creating friction against your fingers as you touch your face. In chemistry, saponification is the normal process used to make soap, but when it happens on your skin without you wanting it to, it quietly strips away your skin’s natural acid mantle and leaves behind a thin, waxy residue. Data from the United States Geological Survey (USGS) and the UK Water Quality Map backs this up — areas with “Very Hard” water (above 180 ppm) consistently show the highest number of people complaining about “moisturizer rejection,” where even expensive creams and serums seem to stop working properly. This points to a clear connection between the mineral buildup from hard water and why so many people feel their topical hydration products are failing them. If you’re dealing with this exact issue and want real, practical ways to deal with hard water skin film.
The Absorption Audit: Why Humectants Fail
If you’ve ever wondered why even the best moisturizers and serums seem to sit on top of your skin instead of sinking in properly in hard water areas, the answer lies in a simple mismatch between your products and that invisible hard water skin film sitting on your face. When you look at the molecular weight of common humectants compared to the dense, waxy layer left behind by hard water, everything starts to make sense. Take hyaluronic acid, for example — it’s supposed to pull water deep into the skin and hold it there, but when that thin hard water skin film is present, the hyaluronic acid molecules get trapped on the surface and can’t penetrate where they’re needed most. The same thing happens with occlusives like petrolatum or shea butter: if you layer them over the mineral residue, you’re basically sealing the hard water film in place, which can lead to localized irritation, clogged pores, and that congested, heavy texture so many people complain about. On top of that, hard water usually comes out with a high pH around 8.5, while your skin barrier works best at a gentle acidic pH of about 5.5. This constant alkaline shift, combined with the mineral buildup, throws off your skin’s delicate lipid bilayers, causing it to lose moisture from the inside while blocking fresh hydration from getting in. For more on how pH changes disrupt the skin barrier lipids, you can check this research on the influence of pH and salts on stratum corneum structure here. It’s a double whammy that leaves your skin feeling tight, dry, and disappointed no matter how carefully you apply your products.
| City / Region | Average Water Hardness (PPM) | Reported Dermal Impact |
| London, UK | 250 – 350 | Persistent “ashy” film, high barrier disruption. |
| San Antonio, TX | 300+ | Significant hard water skin film accumulation. |
| Indianapolis, IN | 280 – 320 | Reported “moisturizer pilling” and texture dullness. |
| Toronto, CAN | 120 – 150 | Moderate mineral buildup on skin. |
Long-Tail Damage: What Hard Water Is Quietly Doing to Your Skin Over the Years
You know, when we talk about hard water damaging skin, most people think it’s just a temporary thing — maybe some tightness after washing. But the real issue is what I call “Long-Tail” damage. This isn’t something that happens after one shower or even one month. It’s the slow, hidden changes that build up over 5, 10, or even 20 years of daily exposure to those minerals in hard water. When that mineral buildup on skin is never properly dealt with, it follows a very specific path that researchers have actually tracked.
It starts with the catalyst effect. Minerals like iron and copper get trapped in that stubborn hard water skin film and, especially when they meet sunlight, they can trigger the Fenton Reaction. This creates harmful free radicals that quietly break down your collagen and elastin fibers year after year, actually speeding up extrinsic aging faster than normal chronological aging alone. Then, over time, the hard water skin film doesn’t just sit on the surface anymore — it starts integrating into the lipid matrix of your skin. Skin biopsies from people in high-mineral areas like London or the Midwestern USA have shown tiny micro-calcifications forming inside the pores. This causes a stiffening of the upper layers, slows down natural cell shedding, and leaves you with that dull, ashy texture that no amount of cream seems to fix.
And finally, there’s the chronic barrier leak. Hard water is strongly alkaline, usually around 8.5 or higher, so years of exposure gradually weaken your skin’s natural acid mantle. Studies on Transepidermal Water Loss (TEWL) show that people living in these “Red Zone” hard water districts lose moisture at a much higher rate. The skin basically becomes “leaky” — constantly losing hydration from the inside while the mineral film blocks new moisture from getting in. As dermatologist Dr. Dennis Gross puts it, many of the impurities in hard water can form free radicals that damage healthy skin cells and lead to a breakdown of collagen. For more details on how hard water affects the skin barrier, you can read this study on the effect of water hardness on surfactant deposition and skin barrier function here.
If you’re dealing with this long-term damage and want to explore ways to support skin repair from the inside out, check out my experiment with PDRN and salmon DNA signaling here.
Why “Double Cleansing” Often Fails to Remove the Film
The trend of “Double Cleansing” has taken over the USA and UK markets, promising a “blank slate” for your serums. However, if you are washing your face with high-TDS tap water, this popular two-step method can actually backfire, physically thickening the hard water skin film instead of dissolving it.
The Anionic Trap: Why More Soap Equals More Scum
Most traditional cleansers rely on anionic surfactants. These molecules carry a negative charge. In a laboratory setting, when these negatively charged molecules meet the cationic (positively charged) minerals like Calcium and Magnesium in your tap water, they bond instantly.
This chemical marriage creates an insoluble curd. Instead of the soap lifting away the dirt, the minerals “grab” the soap and pull it down onto the dermis. This is how you end up with a hard water skin film that feels like a waxy coating. When you “Double Cleanse” with standard products in a hard water zone like London or Chicago, you are performing two rounds of “Saponification” on your own face. You are layering, not cleaning.
The Chelating Reality: Breaking the Ionic Bond
If “Double Cleansing” is failing, it is usually because the products being used lack the “Molecular Magnets” known as chelating agents. Standard surfactants cannot “unlock” a mineral bond once it has formed on the skin.
Only soap-free synthetic detergents or specific chelating formulations (containing ingredients like Disodium EDTA or Sodium Phytate) have the chemical strength to surround a mineral ion and lift it off the skin. Without these specific “Mineral-Lifters,” the hard water skin film remains undisturbed. It continues to act as a plastic-like wrap over your pores, leading to the “Long-Tail” damage—where your expensive PDRN serums or moisturizers simply cannot reach the living cells they are meant to treat.
While mineral buildup on skin creates a physical blockade on the surface, the underlying dermal cells often enter a state of ‘signaling fatigue.’ Research into regenerative molecules suggests that once the hard water skin film is addressed, the skin requires specific cellular cues to repair the fragmented collagen. In our previous technical review, The PDRN Experiment: Analyzing Salmon DNA Signaling, we audited how polynucleotides interact with the skin barrier to jumpstart this recovery process.
Conclusion: Reporting the Environmental Reality
The data gathered in this technical review points to a singular, often overlooked conclusion: the hard water skin film is not a cosmetic myth, but a physical blockade. For those residing in the “Red Zones” of the USA and UK—where mineral concentrations are highest—understanding the chemistry of your tap water is the first step in auditing your dermal health.
Throughout this analysis, we have reported on the chemical facts of saponification, the documented shift in alkaline pH, and the historical archives of product rejection. The evidence suggests that mineral buildup on skin is the primary “invisible” factor in why high-end moisturizers often fail to perform as advertised.
When the skin is coated in a waxy, metallic shield, even the most advanced formulas cannot reach the cells they are intended to nourish. Recognizing this environmental reality allows for a more forensic approach to skincare—one that accounts for the water in your pipes before questioning the quality of the products on your shelf.
Technical FAQ: Hard Water & Dermal Science
The “squeaky” sensation is a physical indicator of hard water skin film. This occurs when minerals like calcium react with your skin’s natural oils to create a waxy residue (calcium stearate). It is not a sign of cleanliness, but of a chemical friction layer.
Yes. Technical audits show that mineral buildup on skin creates a non-permeable shield. This “skin scum” prevents humectants like Hyaluronic Acid from reaching the stratum corneum, causing the product to sit on the surface or “pill.”
Long-term exposure to high-TDS water can lead to chronic barrier degradation. Minerals like Iron act as catalysts for oxidative stress, which has been observed to fragment collagen fibers over years of accumulation.
Not necessarily. If using anionic surfactants with hard water, double cleansing can actually increase the hard water skin film. Standard soaps bond with minerals to create more residue; only chelating agents can effectively break these ionic bonds.
The “ashy” appearance is a light-scattering effect. The microscopic crystals in the hard water skin film prevent light from reflecting evenly off the dermis, resulting in a matte, dull complexion regardless of hydration levels.
Healthy skin maintains an acidic pH of 5.5. Technical data shows hard water often reaches an alkaline pH of 8.5. This shift disorganizes the lipid barrier, leading to increased transepidermal water loss (TEWL).
Technically, yes. The reaction between calcium ions and fatty acids (sebum) is the exact chemical process used to make soap. On the skin, this results in the hard water skin film known as calcium stearate.
Reports suggest this is often due to the mineral salts drying into the pores. As the water evaporates, the minerals remain, potentially causing micro-irritation and disrupting the delicate microbiome balance.
Filters using KDF-55 media are documented to reduce chlorine and some heavy metals. However, unless the filter specifically softens the water or you use a chelating cleanser, some mineral buildup on skin may still occur.
A common indicator is “product rejection”—when moisturizers feel greasy on top but your skin feels tight underneath. This suggests the hard water skin film is blocking absorption.